Accelerate your preclinical discoveries
The Translational Research-Based preclinical CRO.
HUMANA Biosciences overview
Humana Biosciences is a tailored preclinical CRO focused to improve preclinical research efficacy.
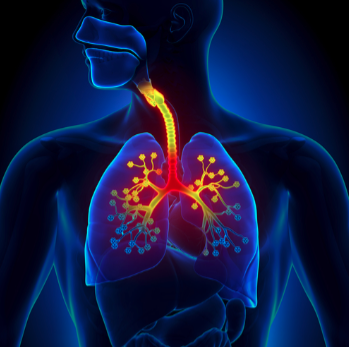
We have a large expertise in Gastroenterology, Urology and Respiratory Pathologies. We also have additional expertise in Dermatology and Nephrology.
We regularly collaborate with Nutraceutical Companies as well as Companies specialized in Medicinal Plant Extracts.
Every study we conduct is a custom designed study tailored specifically for the client. We offer our customers the best predictive experimental models for chronic functional pathologies.
Our mission is to reduce the number of inefficacious candidates moving through to the later stages of clinical investigation by developing innovative experimental models (in vitro and in vivo). Partnering with Humana Biosciences also gives you on-demand access to ethically obtained human tissues.
OUR ULTIMATE AMBITION IS TO MAKE YOUR PRE-CLINICAL DRUG DEVELOPMENT MORE PREDICTIVE AND FASTER.
They trusted us












The Humana Biosciences Experience
Services
Our services are based on the Translational Pharmacology Approach (TPA) with the aim of improving the predictive value of the experimental models used to qualify the efficacy and safety of a drug candidate.
We propose a large choice of experimental platforms:
– Human Tissues from Healthy donors and Patients. Tissues can be fresh, frozen at -80°C or paraffin embedded (FFPE)
– Human primary cells in Air-Liquid Interface (ALI) cultures
– Human and animal isolated tissues freshly isolated for studies of myogenic and neurogenic contractions/relaxations
– Experimental models in vivo on mice and rats, mimicking the pathology of interest
Research Papers
Humana Biosciences was founded by Dr. Stefano Palea, a pharmacologist with over 35 years of experience in preclinical research, particularly in Urology.
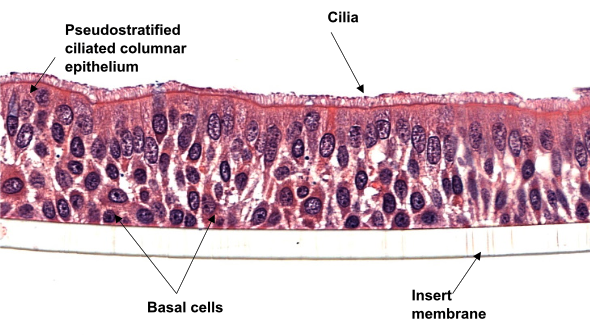
Alongside a highly published Scientific Advisory Board, our expertise spans receptor pharmacology, neuro-urology, cilia analysis, botulinum toxin assays, and innovative drug delivery systems.
Research Projects
Our internal research focuses on four key areas:
- Botulinum toxin potency testing using in vitro, ex vivo and in vivo assays
- 3D cultures of human bronchial cells from healthy and diseased donors
- In vivo models of asthma and COPD in rodents
- Comparative pharmacology on pig and human smooth muscle tissues.
About us
The team is made up of scientists and research technicians who bring years of experience and knowledge into the preclinical research models we perform in our laboratories. Globally, our employees have more than 7 years of internal service, thereby maintaining a low level of employee turnover and guaranteeing technical stability and robustness.
Humana Biosciences Management and Scientific Advisory Board members has combined experience of over 200 years of drug discovery and development know-how, by working in Pharma companies, Biotechs as well as Academic laboratories. Together they published more than 750 papers in peer-reviewed International Journals.
Need to speed up you preclinical research ?
Contact us !
News & Events
Still have a question ?
Question 1
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.